 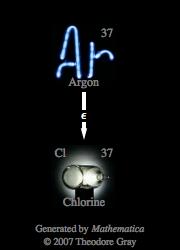  
A positron emission releases a positron. An alpha decay releases an alpha particle. (Note that, just as presented in the equation, a Beta particle has a mass number of 0 and an atomic number -1, kind of similar to an electron). Hence, it is easy and straight forward to see that this radioactive decay is to relaese a Beta particle and the decay is a Beta decay. The changes in atomic and mass numbers is accounted for in the particular particle that that is emitted and subsequently termed the type of radioactive decay that is going on.įor example, this equation represented shows that the Bi atom splits into a Po and a Beta particle. The type of radioactive decay is usually discernable from studying the products and reactants (the parent nucleus/atom, the daughter nucleus and the emitted or absorbed particles) of the radioactive decay. What type of decay is illustrated by the equation below? The type of decay illustrated by the equation is a Beta decay. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
